In prokaryotes, translated peptides are actively pushed through the SecY channel by a protein called SecA. Posttranslational translocation in prokaryotesĪnimation of posttranslational translocation in prokaryotes. In the animation, the structure of BiP is approximated using the structure of hsc70 ( 1YUW), and the J-domain of Sec63 is based on that of auxilin ( 2QWN). 
The SBD of BiP binds non-specifically to the peptide as it enters the ER lumen, and keeps the peptide from sliding backwards in a ratchet-type mechanism. The primary role of the membrane protein complex Sec62/Sec63 is to activate the ATPase activity of BiP via a J-domain located on the lumen-facing portion of Sec63. In the animation, the ATP-bound NBD is shown in purple, and the ADP-bound state is shown in blue. While the NBD is bound to ATP, the SBD is in an open state, allowing for peptide release, while in the ADP state, the SBD is closed and peptide-bound. 
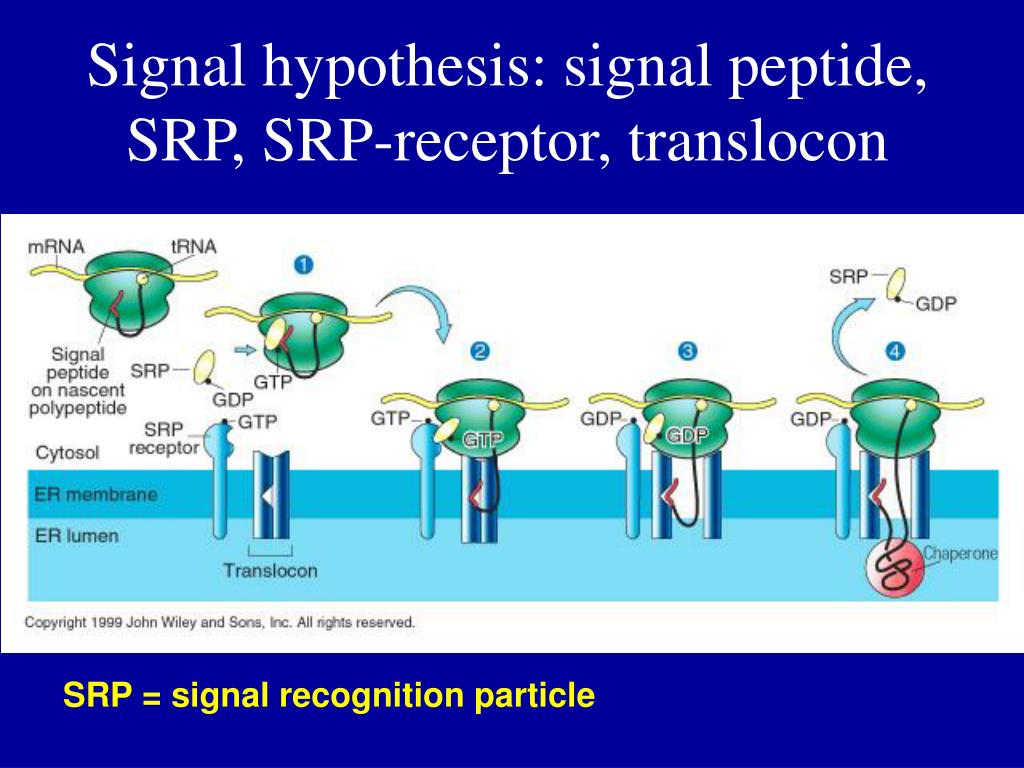
The nucleotide binding state of the NBD determines whether the SBD can bding to a substrate peptide. In eukaryotes, posttranslational translocation requires the Sec62/Sec63 complex (shown in green in the animation) and the chaperone protein BiP (shown in purple/blue).īiP is a member of the Hsp70 family of ATPases, a group which is characterized as having an N-terminal nucleotide-binding domain (NBD), and a C-terminal substrate-binding domain (SBD) which binds to peptides. Eukaryotes and prokaryotes have evolved different mechanisms to ensure the successful translocation of synthesized peptides. During posttranslational translocation, additional proteins are necessary to ensure that the peptide moves unidirectionally into the ER. Posttranslational translocation in eukaryotesĪnimation of posttranslational translocation in eukaryotes.ĭuring cotranslational translocation, the ribosome provides the motive power that pushes the growing peptide into the ER lumen. During translocation, the signal sequence is cleaved by a signal peptide peptidase, freeing the amino terminus of the growing peptide. As shown in the animation, the signal sequence of the nascent peptide intercalates into the walls of the channel, through a side opening known as the lateral gate. The pore of the channel, formed by the α-subunit, is blocked by a short helical segment which is thought to become unstructured during the beginning of protein translocation, allowing the peptide to pass through the channel. The Sec61 translocation channel (known as SecY in prokaryotes) is a highly conserved heterotrimeric complex composed of α-, β- and γ-subunits. As the SRP and SRP receptor dissociate from the ribsome, the ribosome is able to bind directly Sec61. The binding of SRP to its receptor causes the SRP to dissociate from the ribosome, and the SRP and SRP receptor also dissociate from each other following GTP hydrolysis. The signal sequence of the nascent peptide chain is then transferred to the protein channel, Sec61. The ribosome is targeted to the ER membrane through a series of interactions, starting with the binding of the SRP by the SRP receptor. The SRP in eukaryotes is a large ribonucleoprotein which, when bound to the ribosome and the signal sequence of the nascent peptide, is able to arrest protein translation by blocking tRNA entry. Proteins that are targeted for translocation across the ER have a distinctive amino-terminal signal sequence (shown in yellow in the animation) which is recognized by the signal recognition particle (SRP). Animation of eukaryotic cotranslational translocation, with a focus on the conformational changes of the protein channel Sec61.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
